Carboxylic Acid and Acid DerivativesHard
Question
3g of activated charcoal was added to 50 mL of acetic acid solution (0.06N) in a flask. After an hour it was filtered and the strength of the filtrate was found to be 0.042 N. The amount of acetic acid adsorbed (per gram of charcoal) is :
Options
A.42 mg
B.54 mg
C.18 mg
D.36 mg
Solution
∵ Number of moles of CH3COOH adsorbed = (0.06 – 0.042) ×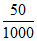
∴ Amount of CH3COOH adsorbed per gram of charcoal =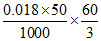 = 0.018 gm
= 0.018 gm
= 18 mg
∴ Amount of CH3COOH adsorbed per gram of charcoal =
= 18 mg
Create a free account to view solution
View Solution FreeTopic: Carboxylic Acid and Acid Derivatives·Practice all Carboxylic Acid and Acid Derivatives questions
More Carboxylic Acid and Acid Derivatives Questions
Name the products in the acid-base reaction:(a) CH3CH2NH2 + HI (b) (CH3)3N + HBr...Reaction of with a mixture of Br2 and KOH gives R - NH2 as the main product. The intermediates. involved in this reactio...Identify Z in the following reaction sequence CH3I X Y Z -...Which compound will liberate CO2 from NaHCO3 solution :...Which gas will be evolved out when [CH3CH2NH2 + (CH3)2CHNH2] is treated with sodium nitrite and HCl :...