Hydrogen and its compoundHard
Question
Calculate the amount of ammonium chloride required to dissolve in 500 ml water to have pH = 4.5 (Kb for NH4OH is 1.8 × 10-5) :-
Options
A.1.8 g
B.0.9 g
C.48.15 g
D.45.15 g
Solution
pH = 7 - 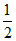 pKb -
pKb - 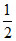 log C
log C
4.5 = 7 -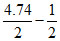 log C
log C
- 0.13 = -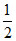 logC
logC
C = 1.80 mol/litre
Amount in 500 mL = 1.80 × 0.5 × 53.5 = 48.15g
4.5 = 7 -
- 0.13 = -
C = 1.80 mol/litre
Amount in 500 mL = 1.80 × 0.5 × 53.5 = 48.15g
Create a free account to view solution
View Solution FreeMore Hydrogen and its compound Questions
A metal M readily forms its sulphate MSO4 which is water - soluble. It forms its oxide MO which becomes inert on heating...Which of the following solution contains approximately equal hydrogen ion concentration....What is the major product expected from the following reaction ?Where D is an isotope of Hydrogen....A metal which is soluble in both water and liquid NH3 separately -...Oxide of an element A is used in the manufacturing fire clay bricks and this A not react with water but its oxides disso...