Analytical ChemistryHard
Question
Dimethyl gloxime gives a red preipitate with Ni2+, which is used for its detection. To get this precipitate reddily, the best pH range is :
Options
A.< 1
B.2 - 3
C.3 - 4
D.9 - 11
Solution
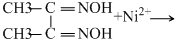
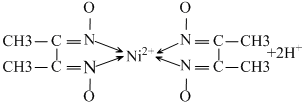
The reaction indicates that protons are released during reaction. Hence a basic medium is needed to facilitate the reaction. Thus the best pHrange is 9 - 11. If medium is kept acidic then nitrogen atom of the ligand will start donating lone pair to H+ ion of the acid and not to central metal ion.
Create a free account to view solution
View Solution FreeMore Analytical Chemistry Questions
An aqueous solution of compound ′A′ gives white precipitate with 2M HCl. The precipitate becomes black on ad...Cu2+ ions give white precipitate with :...Which of the following cations form(s) black precipitate(s) with H2S (g) ?...Which of the following is insoluble in dil. HNO3 but dissolves in aquaregia ?...Which of the following is/are correct for potassium ferrocyanide ?...