Coordination CompoundHard
Question
Which of the following metal ions will form complexes with the same magnetic momentand geometry irrespective of the nature of ligands?
Options
A.Ni2+
B.Fe2+
C.Cu2+
D.Co2+
Solution
Cu2+ forms complexes with the same magnetic moment and geometry irrespective of the nature of ligand. It can be explained by electronic configuration.
Cu2+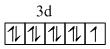
It has 9 electrons in 3d shell. Hence any incoming ligand whether it is strong or weak will result imn the formation of same. geometry (if number of ligands is same) because any ligand can do nothing with this umpaired e- in 3d shell. Moreover, the complex formed will have same magetic moment due to this unpaired e- .
Cu2+
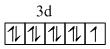
It has 9 electrons in 3d shell. Hence any incoming ligand whether it is strong or weak will result imn the formation of same. geometry (if number of ligands is same) because any ligand can do nothing with this umpaired e- in 3d shell. Moreover, the complex formed will have same magetic moment due to this unpaired e- .
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
Which of the following is considered to be an anticancer species?...Which of the following will have two stereoisomeric forms?...The complexes [Co (NH3 )6] [Cr (CN )6 ] and [Cr (NH3 )6 ] [Co (CN)3 ] are the examples of which type of isomerism ?...CoCl3.4H2O is an anhydrous binary solute hence its Werner′s representation is :...A co-ordination complex has the formula PtCl4.2KCl. Electrical conductance measurements indicate the presence of three i...