Coordination CompoundHard
Question
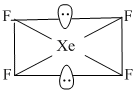
Shape of XeF4 molecule is :
Options
A.Pyramidal
B.Square planar
C.Triangular planar
D.Linear
Solution
Shape of XeF4 molecule is square planner. It involves sp3d2 hybridisation.

Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
Coordination compounds [Pt(NH3)3(NCS)] and [Pt(NH3)3(SCN)] are examples of......isomerism...In which of the following coordination entities, the magnitude of ∆0 [CFSE in octahedral field] will be maximum?:...Ethylene diamine is an example of a .......... ligand :...Effective atomic number of Co(CO)4 is 35, hence it is less stable. It attains stability by...What is the charge on the complex [Cr(C2O4)2(H2O)2] formed by Cr(III) ?...