ThermochemistryHard
Question
What will be the heat of formation of methane, if the heat of combustion of carbon is′-x′ kJ, heat of formation of water is′-y′ kJ and heat of combustion of methane is′z′ kJ ?
Options
A.(- x - y - z)kJ
B.(- z - x + 2y)kJ
C.(- x - 2y - z) kJ
D.(- x - 2y + z)kJ
Solution
From given data, we have
C + O2 → CO2 - x kJ ......(i)
H2 +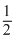 O2
O2 → H2O - y kJ ......(ii)
CH4 + 2O2 → CO2 + H2O + z kJ ......(iii)
The required equation is
C + 2H2 →CH4 + Q
To get the requred equation, poerate (i) + 2 × (ii) - (iii)
Thus, we get
C + 2H2 → CH4 + [(-x) + (-2y) - (-z)]
Thus, heat of formation of methane is (- x - 2y = z) kJ
C + O2
H2 +
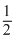 O2
O2 CH4 + 2O2 → CO2 + H2O + z kJ ......(iii)
The required equation is
C + 2H2
To get the requred equation, poerate (i) + 2 × (ii) - (iii)
Thus, we get
C + 2H2
Thus, heat of formation of methane is (- x - 2y = z) kJ
Create a free account to view solution
View Solution FreeMore Thermochemistry Questions
From the following data at 25o C, which of the following statement(s) is/are correct?½H2(g) + ½O2(g)→ OH(g): ΔHo = 42 kJ...Given the following equations and ᐃHo values, determine the enthalpy of reaction at 298 K for the reaction : C2H4 ...Given thatC(s) + O2(g) → CO2(g); ΔHo = −X kJ2CO(g) + O2(g) → 2CO2(g): ΔHo = −Y kJThe enthalpy of formation of carbon mon...Which of the following salts shall cause more cooling when one mole of the salt is dissolved in the same amount of water...For the following reaction, C6H12O6(s) + 6O2(g) → 6CO2(g) + 6H2O(l)Which of the following expression is incorrect?...