ThermochemistryHard
Question
ᐃHfo(298K) of methanol is given by the chemical equation :
Options
A.CH4(g) + 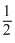 O2(g) → CH3OH(g)
O2(g) → CH3OH(g)
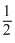 O2(g) → CH3OH(g)
O2(g) → CH3OH(g)B.C(graphite) + 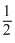 O2(g) + 2H2(g) → CH3OH(l)
O2(g) + 2H2(g) → CH3OH(l)
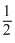 O2(g) + 2H2(g) → CH3OH(l)
O2(g) + 2H2(g) → CH3OH(l)C.C(diamond) + 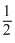 O2(g) + 2H2(g) → CH3OH(l)
O2(g) + 2H2(g) → CH3OH(l)
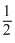 O2(g) + 2H2(g) → CH3OH(l)
O2(g) + 2H2(g) → CH3OH(l)D.CO(g) + 2H2(g) → CH3OH(l)
Solution
Heat of formation is defined as the heat exchange when one mole of a compound isformed from its constituent elements at STP. In case of allotrope, most stable allotrope is taken of allotrope, most stable allotrope is taken into account. Among graphite and diamond; graphite is stabler. hence, heat of formation of CH3OH represented by the eqn.
C(graphite) +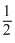 O2(g) + 2H2(g) → CH3OH(l)
O2(g) + 2H2(g) → CH3OH(l)
C(graphite) +
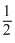 O2(g) + 2H2(g) → CH3OH(l)
O2(g) + 2H2(g) → CH3OH(l)Create a free account to view solution
View Solution FreeMore Thermochemistry Questions
The enthalpy change for the following process would be expected to be a negative for...The standard enthalpies of formation of NH3(g), CuO(s) and H2O(l) are −46, −155 and −285 kJ/mol, respectively. The entha...Benzene burns in oxygen according to the following reactions.C6H6(l) + 15/2 O2(g) → 3H2O(l) + 6CO2(g)If the standard ent...Given that :-CH4(g) + 300 kJ → C(g) + 4H(g)C2H6(g) + 620 kJ → 2C(g) + 6H(g)The value of C - C bond energy is...The enthalpy of combustion of propane (C3H8) gas in terms of given data is : Bond energy (kJ/mol) εC-H εO=O &#...