ThermochemistryHard
Question
The heat of combustion of yellow phosphorous is - 9.91 kJ and of red phosphorous to redphosphorus is :
Options
A.- 9.91 kJ
B.- 8.78 kJ
C.- 9.34 kJ
D.- 1.13 kJ
Solution
P(red) + 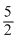 O2 → P2O5 ; H = - 8.78
O2 → P2O5 ; H = - 8.78
P(yellow) +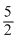 O2 → P2O5 ; = - 9.91
O2 → P2O5 ; = - 9.91
Subtracting
P(red) +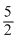 O2 - P(Yellow) = 0;
O2 - P(Yellow) = 0;
ᐃH = - 8.78 + 9.91 = 3.113
P(Red) → P(Yellow) ᐃH = 1.13
P(Yellow) → P(Red) ᐃH = - 1.13
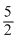 O2 → P2O5 ; H = - 8.78
O2 → P2O5 ; H = - 8.78P(yellow) +
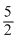 O2 → P2O5 ; = - 9.91
O2 → P2O5 ; = - 9.91Subtracting
P(red) +
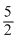 O2 - P(Yellow) = 0;
O2 - P(Yellow) = 0;ᐃH = - 8.78 + 9.91 = 3.113
P(Red) → P(Yellow) ᐃH = 1.13
P(Yellow) → P(Red) ᐃH = - 1.13
Create a free account to view solution
View Solution FreeMore Thermochemistry Questions
Butane exists in various conformations in nature. At any given instant, the probability that a given butane molecule is ...Which of the following molecules will have different values of standard molar enthalpy of formation, one calculated usin...Two bars of different metals are heated to 60ºC and then immersed in identical insulated containers each containing 200 ...Equal volumes of one molar hydrochloric acid and one molar sulphuric acid are neutralized completely by dilute NaOH solu...The molar enthalpy of vaporization of benzene at its boiling point (353 K) is 7.4 kcal/mol. The molar internal energy ch...