ThermochemistryHard
Question
S + O2 + x kcal .......(1)
SO2 +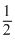 O2 → SO2 + y kcal .......(2)
O2 → SO2 + y kcal .......(2)
The heat of formation of SO3 in the above reactior :
SO2 +
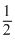 O2 → SO2 + y kcal .......(2)
O2 → SO2 + y kcal .......(2)The heat of formation of SO3 in the above reactior :
Options
A.(x + y)
B.(x - y)
C.(2x - y)
D.(2x + y)
Solution
S = O2 → SO2 = x ......(i)
SO2 +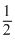 O2 → SO3 + y ......(ii)
O2 → SO3 + y ......(ii)
Adding (i) and (ii) we have
S + O2 + SO2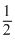 O2 → S O2 + S O3 + y
O2 → S O2 + S O3 + y
s +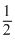 O2 → SO3
O2 → SO3
So, heat of information of SO3 will be (x - y).
SO2 +
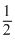 O2 → SO3 + y ......(ii)
O2 → SO3 + y ......(ii)Adding (i) and (ii) we have
S + O2 + SO2
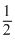 O2 → S O2 + S O3 + y
O2 → S O2 + S O3 + ys +
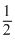 O2 → SO3
O2 → SO3So, heat of information of SO3 will be (x - y).
Create a free account to view solution
View Solution FreeMore Thermochemistry Questions
A + BA gives iodoform test. ′B′ is heated with excess of H-I. then ′C′ is obtained A and C are :...ΔH of which of the following reaction(s) may be directly taken as enthalpy of combustion of the concerned substance?...The thermochemical equation for the dissociation of hydrogen gas into atoms may be written asH2 → 2H; ΔH = 432.0 kJ.What...Which of the following are true about resonance energy ?...Which of the following is arranged in the increasing order of enthalpy of vaporisation ?...