Ionic EquilibriumHard
Question
24. Air containing 79% of nitrogen and 21% of oxygen by volume is heated at 2200K and 1atm util equilibrium is establised accoeding to the reaction N2(g) + O2(g) ⇋ 2NO(g)
If the Kp of the raection is 1.1 × 10-3 calculate the amount of nitric oxide produced in terms of volume percent.
If the Kp of the raection is 1.1 × 10-3 calculate the amount of nitric oxide produced in terms of volume percent.
Options
A.1.33
B.1.12
C.1.02
D.1.44
Solution
N2(g) + Q2(g) ⇋ 2NO(g)
At equilibrium, we have [N2] = 0.79(1 - α); [O2] = 0.21(1 - α); [NO] = 2α
Total number of moles
= 0.79(1 - α) + 0.21(1 - α) + 2a = 1 + α
PN2 =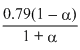 × 1;
× 1;
PO2 =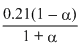 × 1; pNO =
× 1; pNO = 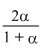 × 1
× 1
Kp =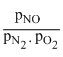
1.1 × 10-3 =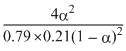
or α = 0.0067
⇒ vol % of NO = 2α × 100
= 2 × 0.0067 × 100 = 1.33%
At equilibrium, we have [N2] = 0.79(1 - α); [O2] = 0.21(1 - α); [NO] = 2α
Total number of moles
= 0.79(1 - α) + 0.21(1 - α) + 2a = 1 + α
PN2 =
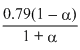 × 1;
× 1;PO2 =
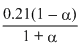 × 1; pNO =
× 1; pNO = 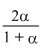 × 1
× 1Kp =
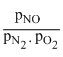
1.1 × 10-3 =
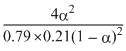
or α = 0.0067
⇒ vol % of NO = 2α × 100
= 2 × 0.0067 × 100 = 1.33%
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
The pH value of 0.1 M solutions of CH3COONa (I), CH3COOH (II), CH3COONH4 (III), NaOH (IV) and HCl (V) is in the order...From separate solutions of for sodium salts NaW, NaX, NaY and NaZ has pH 7.0, 9.0 10.0 and 11.0 respectively. When each ...The Ksp of Ag2CrO4 = 1.2 × 10–11. What concentration of Ag+ ion in aqueous solution will just fail to give a precipitate...In which of the following combinations, is buffer action expected?1. NH3 + NH4CI in 1 : 1 mole ratio2. HCI + NaCI in 1 :...Ksp of AgCl is 1.96 × 10-10. 100 mL of saturated AgCl solution is titrated with 1 × 10-5 M NH4 SCN. Volume of ...