Analytical ChemistryHard
Question
Compound (X) when heated with KOH solution it produce colorless poisonous gas (Y) having rotten fish smell and solution (Z).
CuSO4(sol.) 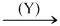 Black ppt (P1)
Black ppt (P1)
CuSO4(sol.) 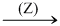 White ppt (P2) + Orange yellow solution (S).
White ppt (P2) + Orange yellow solution (S).
Select the correct statements :-
Options
A.Gas (Y) is hydrogen sulphide
B.Oxidation number of metal is +1 in white ppt (P2)
C.Compound (X) contain NH4+ ion
D.Orange yellow solution (S) is decolourised on addition of sodium thiosulphate
More Analytical Chemistry Questions
Which one of the following metal salts produces a blue coloured bead in cobalt nitrate charcoal cavity test ?...CrO42- + H+ + H2O2 X + H2OIdentify the correct statement with respect to X....Sometimes yellow turbidity appears while passing H2S gas even in slightly acidic medium the absence of II group radicals...Ammonium molybdate test is used for the estimation of :...Consider three metal chlorides $x,y$ and z , where x is water soluble at room temperature, y is sparingly soluble in wat...