ThermochemistryHard
Question
AB, A2 and B2 are diatomic molcules. If the bond enthalpies of A2 AB and B2 are in the ratio 1 : 1 :0.5 and enthalpy of formation of AB from A2 and B2 is - 100kJ mol-1. What is the bond energy of A2
Options
A.200 kJ mol-1
B.100 kJ mol-1
C.300 kJ mol-1
D.400 kJ mol-1
Solution
Let bond energy of A2 be x than bond energy of AB is also x and bond energy of B2 is x/2.
Enthalpy of formation of AB is - 100 kJ/mole : A2 + B2 → 2AB
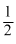 A2 +
A2 + 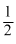 B2 → 2AB; ᐃH = - 100kJ
B2 → 2AB; ᐃH = - 100kJ
or - 100 =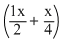 - x
- x
∴ - 100 =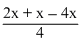 ∴ x = 400 kJ
∴ x = 400 kJ
Enthalpy of formation of AB is - 100 kJ/mole : A2 + B2 → 2AB
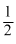 A2 +
A2 + 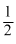 B2 → 2AB; ᐃH = - 100kJ
B2 → 2AB; ᐃH = - 100kJor - 100 =
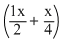 - x
- x∴ - 100 =
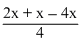 ∴ x = 400 kJ
∴ x = 400 kJCreate a free account to view solution
View Solution FreeMore Thermochemistry Questions
Using the data provided, calculate the multiple bond energy (kJ mol-1) of a C ≡ C bond in C2H2. That energy is (take t...At 300 K, the standard enthalpies of formation of C6H5COOH(s), CO2(g) and H2O(l) are −408, −393 and −286 kJ/mol, respect...A heat engine is operating between 500K to 400K. If the engine absorbs 100J heat, then which of the following is impossi...When 1.0 g of oxalic acid (H2C2O4) is burned in a bomb calorimeter whose heat capacity is 8.75 kJ/K, the temperature inc...The standard enthalpy of formation of a substance...