Gaseous StateHard
Question
X ml of H2 gas effuse thruogh a hole in a container in 5 seconds.
The time taken for effusion of the same volume of the gas specified below underindentical condition is
The time taken for effusion of the same volume of the gas specified below underindentical condition is
Options
A.10 seconds : He
B.20 secunds : O2
C.25 seconds : CO
D.55 seconds : CO2
Solution
For effusion of same volume,
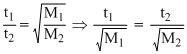
This clearly seen from the option that the ratio of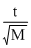 is same for H2 and O2
is same for H2 and O2
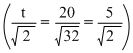
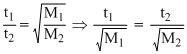
This clearly seen from the option that the ratio of
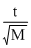 is same for H2 and O2
is same for H2 and O2 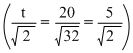
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
Temperature at which r.m.s. speed of O2 is equal to that of neon at 300 K is :...For one mole of a van der Waals gas when b = 0 and T = 300 K, the PV vs. 1/V plot is shown below. The value of the van d...One mole of a non-ideal gas undergoes a change of state (2.0 atm, 3.0L, 95K) → (4.0 atm, 5.0L, 245K) with a change...Which nobal gas has maximum solubility in water :-...Which is not correct about the Schlocky defects?...