Chemical BondingHard
Question
Which of the following has the lowest diploe moment ?
Options
A.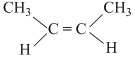
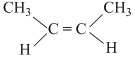
B.CH3C ≡ C CH3
C.CH3CH2C ≡ CH
D.CH2 = CH - C ≡ CH
Solution
CH3C ≡ CCH3 being symmetrical, has the lowest dipole moment
Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
For the cell given below ECell - Eocell = - 0.118 V Zn/Zn2+ || Cu2+/CuWhat is the ratio of [Zn2+]/[Cu+2] :-...Match the polymers in column-A with their main used in column-B and choose the correct answer :Column-A Column-B(A) Poly...Which of the following statements is/are wrong ?The following reactions occur:i. P4 + 5O2 → P4O10.ii. P4 + 3O2 ...Which shows largest bond angle :-...CO2 has the same geometry as :-(a) HgCl2 (b) NO2(c) SnCl2 (d) C2H2...