ElectrochemistryHard
Question
The products formed when an aqueous solution of NaBr is electrolysed in a cell having electrodes are :
Options
A.Na and Br2
B.Na and O2
C.H2, Br2 and NaOH
D.H2 and O2
Solution
NaBr ⇋ Na+ + Br-
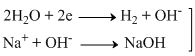 At cathode
At cathode
Br- → Br + e-
Br + Br → Br2
So the products are H2 and NaOH (at cathide) and Br2 (at anode)
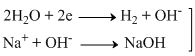 At cathode
At cathodeBr- → Br + e-
Br + Br → Br2
So the products are H2 and NaOH (at cathide) and Br2 (at anode)
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
For the reduction of silver ions with copper metal, the standard cell potential was found to be +0.46 V at 25oC. The val...Assertion :- BeSO4 is more soluble in water than BaSO4.Reason :- In BeSO4 H.E. exceeds its L.E....Saturated solution of KNO3 is used to make ‘salt bridge’ because...In hydrogen - oxygen fuel cell, combustion of hydrogen occurs to...If a current of 1.0 A is drawn from the Daniel cell for 96.5 min, the cathode will gain in weight by (Cu = 63.5, Zn = 65...