Chemical BondingHard
Question
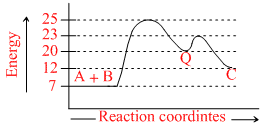
In a multistep reaction such as A + B → Q → C. The potential energy diagram is shown below. What is Ea for the reaction Q → C ?

Options
A.3 kcal mol-1
B.5 kcal mol-1
C.8 kcal mol-1
D.11 kcal mol-1
Solution
For Q → C.
Ea = 23 - 20 = 3 kcal mol-1
Ea = 23 - 20 = 3 kcal mol-1
Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
The difference between amylose and amylopectin is...Amongst LiCl, RbCl BeCl2 and MgCl2, the compounds with the greatest and the least jonic character, respectively are :-...Which of the following structure is similar to graphite?...Incorrect order according to the property is :...The molecule which has zero dipole moment is :...