Atomic StructureHard
Question
The uncertainties in the velocities of two particle, A and B are 0.05 and 0.02 ms-1 respectively. The mass of B is five time to that of the mass of A. What is the ratio of uncertain ties 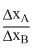 in their position ?
in their position ?
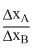 in their position ?
in their position ?Options
A.2
B.0.25
C.4
D.1
Solution
Using the relation,
ᐃx.ᐃv =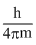 [Heisenberg uncertainty principle]
[Heisenberg uncertainty principle]
or ᐃx =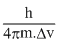
Thus, ᐃxA =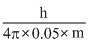 ......(i)
......(i)
ᐃxB =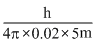 .......(ii)
.......(ii)
Dividing (i) by (ii), we get
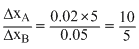 or 2
or 2
ᐃx.ᐃv =
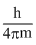 [Heisenberg uncertainty principle]
[Heisenberg uncertainty principle]or ᐃx =
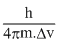
Thus, ᐃxA =
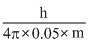 ......(i)
......(i)ᐃxB =
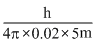 .......(ii)
.......(ii)Dividing (i) by (ii), we get
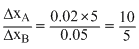 or 2
or 2 Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
The triad of nuclei that is isotonic is...The radius of the hydrogen atom in its ground state is 5.3 × 10–11 m. After collision with an electron it is found to ha...The possible set of quantum numbers for which n = 4, l = 3 and s = + 1/2 is...A proton and an a-particle are accelerated through the same potential difference from rest. Then the ratio of their de B...The wavelength of a charged particle ____the square root of the potential difference through which it is accelerated :...