Atomic StructureHard
Question
The energy of electron in first energy level is - 21.79 × 10-12 erg per atom. The energy of electron in second energy level is :
Options
A.- 54.47 × 10-12 erg atom-1
B.- 5.447 × 10-12 erg atom-1
C.- 0.5447 × 10-12 erg atom-1
D.- 0.05447 × 10-12 erg atom-1
Solution
For a particular element,
En =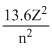 erg atm-1
erg atm-1
∴ En ∝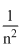
∴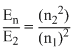
or E2 =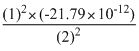
- 5.447 × 10-12 erg atm-1
En =
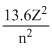 erg atm-1
erg atm-1∴ En ∝
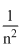
∴
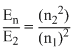
or E2 =
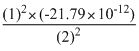
- 5.447 × 10-12 erg atm-1
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
Which of the the following has maximum energy ?...Which of the following element will have the same number of electrons in s and as well as p-type of orbitals?...What is the wave number of the radiation of lowest frequency in the Lyman series of the spectrum of Li2+ ion?...If an electron is revolving around the nucleus of He+ ion at a distance of 4.0 Å, the magnitude of centripetal force on ...The wavelength of a spectral lime for an electronic transition is inverssly related to :...