SolutionHard
Question
Exactly 1g of urea dissolved in 75 g of water give a solution that boils at 100.114oC at 760 torr. The molecular weight of urea is 60.1. The boiling point elevation constant for water is
Options
A.1.02
B.0.51
C.3.06
D.1.51
Solution
Weight of solute (w) = 1 g
Weight of solvent (W) = 75 g
Boling point of solution = 100. 114oC
Boling point of solvent = 100oC
ᐃT = 100.114 -100 = 0.114oC
Molecular weight of solute (m) = 60.1
Boling point elevation caostant (K) = ?
m =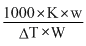
K =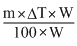
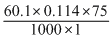
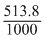 = 0.513
= 0.513
Weight of solvent (W) = 75 g
Boling point of solution = 100. 114oC
Boling point of solvent = 100oC
ᐃT = 100.114 -100 = 0.114oC
Molecular weight of solute (m) = 60.1
Boling point elevation caostant (K) = ?
m =
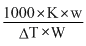
K =
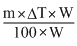
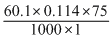
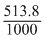 = 0.513
= 0.513Create a free account to view solution
View Solution FreeMore Solution Questions
The solubility of solid silver chromate, Ag2CrO4 is determined in three solvents. (Ksp = 9 × 10-12)(i) Pure water (...Thevan′t Hoff factor, i for a compound which undergoes dissociation in one solvent and association in other solven...PH3 (Phosphine) when passed in aqueous solution of CuSO4 it produce -...One mole of a solute A is dissolved in a given volume of a solvent. The association of the solute take place as follows ...The latent heat of vaporization of a liquid of molar mass, 80 g/mol and boiling point, 127°C is 8 kcal/mol. The ebullios...