SolutionHard
Question
40 ml of 0.1 M ammonia solution is mixed with 20 ml of 0.1 M HCl. What is pH of themixture ?( pkb of ammonia solution is 4.74).
Options
A.4.74
B.2.26
C.9.26
D.5.00
Solution
NH3 + HCl → NH4Cl
pOH = pKb + log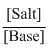
pOH = 4.74 + log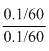
pOH = 4.74 + 0 = 4.74
pOH = 14 - 4.74 = 9.26
pOH = pKb + log
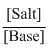
pOH = 4.74 + log
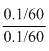
pOH = 4.74 + 0 = 4.74
pOH = 14 - 4.74 = 9.26
Create a free account to view solution
View Solution FreeMore Solution Questions
If pKa = - log Ka = 4, and Ka = Cx2 then Van′t Hoff factor for weak monobasic acid when C = 0.01 M is :...If P0 and P are the vapour pressures of a solvent and its solution respectively and N1 and N2 are the mole fractions of ...pH of 0.02 M aq. solution of NH4Cl (Pkb = 4.73) is :-...A non-volatile solute ‘X’ completely dimerizes in water, if the temperature is below −3.72°C and the solute completely d...Identify the false statement :...