Mole ConceptHard
Question
The reaction of calcium with water is represented by the equation
Ca + 2H2O → Ca(OH)2 + H2
What volume of H2 at STP woild be liberated when 8 gm of calcium completely reacts with water ?
Options
A.0.2cm3
B.0.4cm3
C.2240 cm3
D.4480 cm3
Solution
Ca + 2H2O → Ca(OH)2 + H2
According to the stoichiometry of reaction, 40 gm of Ca on complate reason with water librates = 2 gm H2
∴ 8gm of Ca. on complete reaction with water liberates =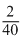 × 8 gm H2
× 8 gm H2
= 0.40 gm H2
=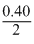 × 22400 cm2
× 22400 cm2
= 4480cm3 of H2 at S.T.P.
According to the stoichiometry of reaction, 40 gm of Ca on complate reason with water librates = 2 gm H2
∴ 8gm of Ca. on complete reaction with water liberates =
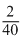 × 8 gm H2
× 8 gm H2= 0.40 gm H2
=
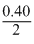 × 22400 cm2
× 22400 cm2= 4480cm3 of H2 at S.T.P.
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
V1 ml of unknown gas (A) + V2 ml of O2 → (V1 + V2) ml of CO2. Gas ‘A’ may be...If a definite volume of ‘20 vol’ H2O2 solution is diluted such that the volume of diluted solution becomes double than t...A person needs an average of 2.0 mg of riboflavin (vitamin B 2 ) per day. How many grams of butter should be taken by th...When a sample of hydrogen fluoride is cooled to 303 K, most of the molecules undergo dimerization. If the vapour density...The commonly used pain reliever, aspirin, has the molecular formula C9H8O4. If a sample of aspirin contains 0.968 g of c...