AIPMT | 2014Chemical BondingHard
Question
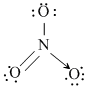
Which one of the following species has plane triangular shape?
Options
A.N3-
B.NO3-
C.NO2-
D.CO2
More Chemical Bonding Questions
The order of decreasing nucleophilicities of the following species is :...The molecule having smallest bond angle is :-...For the following equilibrium H2(g) ⇋ 2H(g)There is 0.1% dissociation at equilibrium pressure of 1 atm. if pressur...The following compounds have been arranged in order of their increasing thermal stability ties. Identify the current ord...Which of the following statements is/are correct ?...