Chemical BondingHard
Question
Which of the following statements is/are correct ?
Options
A.CaC2 is made in an electric furnace by the reaction:
CaO + C → CaC2 + CO.
16.0 g of CaC2 is obtained from 9.0 g of C.
CaO + C → CaC2 + CO.
16.0 g of CaC2 is obtained from 9.0 g of C.
B.Polyethene can be produced from CaC2 as follows.
CaC2 + H2O → CaO + HC ≡ CH
HC ≡ CH + H2 → H2C = CH2
n(CH2 = CH2) → -(CH2 - CH2)n- (polyethene)
32.0 kg of CaC2 producs 14.0 kg of polyethene.
CaC2 + H2O → CaO + HC ≡ CH
HC ≡ CH + H2 → H2C = CH2
n(CH2 = CH2) → -(CH2 - CH2)n- (polyethene)
32.0 kg of CaC2 producs 14.0 kg of polyethene.
C.1.435 g of AgCl is obtained from 17.75 g of [Ag(NH3)2]Cl by the following reaction:
[Ag(NH3)2]Cl + 2HNO3 → AgCl + 2NH4NO3.
[Ag(NH3)2]Cl + 2HNO3 → AgCl + 2NH4NO3.
D.Commercial sodium ′hydrosulfite′ is 50% pure Na2S2O4. It is prepard as follows:
i. Zn + 2SO2 → ZnS2O4
ii. ZnS2O4 + Na2CO3 → ZnCO3 + Na2S2O4
174.0 metric ton of commercial product (Na2S2O4) can be made from 65.4 metric ton of Zn, with a sufficient supply of other reactants.
i. Zn + 2SO2 → ZnS2O4
ii. ZnS2O4 + Na2CO3 → ZnCO3 + Na2S2O4
174.0 metric ton of commercial product (Na2S2O4) can be made from 65.4 metric ton of Zn, with a sufficient supply of other reactants.
Solution
a. CaO + 3C → CaC2 + CO
1 mole 3mol (1 mol = 64 g)
Moles of CaC2 =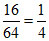 mol
mol
Weight of C = 3 ×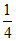 × 12 = 9.0 g
× 12 = 9.0 g
b. Weight of C2H4 = (32.0 kg)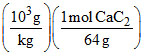
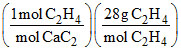
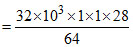
Hence, (c) is wrong
d. (Atomic weight of Zn = 65.4 g, Mw of Na2S2O4 = 174)
Weight of pure Na2S2O4 = (65.4 × 106 g Zn)
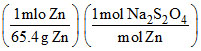
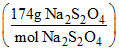
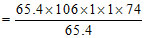
= 174 × 106 g
= 174 reaction ton
Weight of 50% pure Na2S2O4 =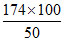
= 348 metric ton
Hence, (d) is wrong.
1 mole 3mol (1 mol = 64 g)
Moles of CaC2 =
Weight of C = 3 ×
b. Weight of C2H4 = (32.0 kg)
Hence, (c) is wrong
d. (Atomic weight of Zn = 65.4 g, Mw of Na2S2O4 = 174)
Weight of pure Na2S2O4 = (65.4 × 106 g Zn)
= 174 × 106 g
= 174 reaction ton
Weight of 50% pure Na2S2O4 =
= 348 metric ton
Hence, (d) is wrong.
Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
Which of the following is correct :-...The species having bond order different from that in CO is :-...Given below are two statements:Statement I: The number of species among ${BF}_{4}^{-},{SiF}_{4},{XeF}_{4}$ and ${SF}_{4}...The ion that is isorlrctronic with CO is :...Using MO theory predict which of the following species has the shortest bond length ?...