ThermodynamicsHard
Question
A thermodynamic system undergoes cyclic process ABCDA as shown in figure. The work done by the system in the cycle is
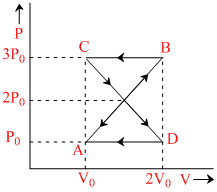

Options
A.P0V0
B.2P0V0
C.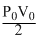
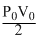
D.Zero
Solution
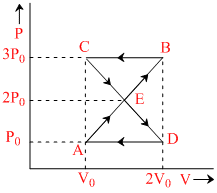
W = Area of BCE + Area of ADE
= - W0 + W0 = 0
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
The amount of work done in converting 1 gm ice at -10o C into steam at 100o C will be : -...An ideal gas is taken around the cycle ABCA as shown in the P - V diagram. The net work done by the gas during the cycle...For isometric process : -...Heat travels through vacuum by :...An ideal gas is taken through the cycle A → B → C → A, as shown in the figure. If the net heat supplie...