Coordination CompoundHard
Question
Which one of the following is an outer orbital complex and exhibits paramagnetic behaviour?
Options
A.[Co(NH3)6]3+
B.[Ni(NH3)6]2+
C.[Zn(NH3)6]2+
D.[Cr(NH3)6]3+
Solution
[Ni(NH3)6]2+
Here in Ni2+ the configuration is
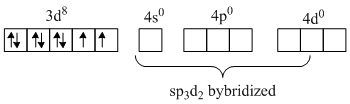
Hence it gives outer orbital octahedral complex which is paramagnetic
Here in Ni2+ the configuration is

Hence it gives outer orbital octahedral complex which is paramagnetic
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
The magnitude of crystal field stabilisation energy in octahedral field depends on I: the nature of the ligand II: the c...The oxidation state of Cr in [Cr(NH3)4Cl2]+ is...Which of the following statements is not correct?...Which of the following statement(s) is /are correct ?...The formula of the complex hydridotrimethoxidoborate (III) ion is:...