Coordination CompoundHard
Question
Which of the following statement(s) is /are correct ?
Options
A.[Co(NH3)6]3+, [Co(CN)6]3- and [Co(NO2)6]3- are diamagnetic involving d2sp3 hybridisation.
B.[Zn(NH3)4]2+, [FeCl4]- and [Ni (CO)4] are diamagnetic involving sp3 hybridisation.
C.The magnetic moment of [Fe(H2O)6]3+ is 5.92 B.M and that of [Fe(CN)6]3- is 1.73.
D.The magnetic moment of K4[MnF6] and K3[FeF6] are same.
Solution
(A) All are diamagnetic and inner orbital complexes because all ligands are strong field ligands.
(B) Fe3+ n = 5 ; μ =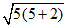 ≈ 5.92 with H2O, so [FeCl4]- is diamagnetic.
≈ 5.92 with H2O, so [FeCl4]- is diamagnetic.
(C) Fe3+ n = 5 ; μ =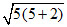 ≈ 5.92 with H2O
≈ 5.92 with H2O
Fe3+ n = 1 ; m = √3 = 1.73 with CN-
(D) Mn2+ (3d5) and Fe3+ (3d5) have same number of unpaired electrons i.e. 5 with weak field ligands, F-
(B) Fe3+ n = 5 ; μ =
(C) Fe3+ n = 5 ; μ =
Fe3+ n = 1 ; m = √3 = 1.73 with CN-
(D) Mn2+ (3d5) and Fe3+ (3d5) have same number of unpaired electrons i.e. 5 with weak field ligands, F-
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
The compound Na2IrCl6 reacts with triphenylphosphine in diethyleneglycol in an atmosphere of CO to give [IrCl(CO)(PPh3)2...Trioxalato aluminate (III) and tetrafluoro-borate (III) ions are respectively...The magnitude of crystal field stabilisation energy in octahedral field depends on I: the nature of the ligand II: the c...The number of unpaired electrons present in complex ion [FeF6]3- is :...As per IUPAC nomenclature, the name of the complex [Co(H2O)4(NH3)2]Cl3 is...