JEE Advanced | 2014d and f-Block ElementsHard
Question
Upon heating with Cu2S, the reagent(s) that give copper metal is/are
Options
A.CuFeS2
B.CuO
C.Cu2O
D.CuSO4
Solution
Cu2S + 2Cu2O 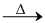 6Cu + SO2
6Cu + SO2
Since, CuSO4 as well as CuO both on heating produces Cu2O as shown below so, they also produce Cu on heating with Cu2S.
2CuO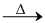 Cu2O +
Cu2O + 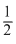 O2
O2
CuSO4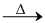 CuO + SO2 +
CuO + SO2 + 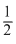 O2
O2
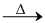 6Cu + SO2
6Cu + SO2Since, CuSO4 as well as CuO both on heating produces Cu2O as shown below so, they also produce Cu on heating with Cu2S.
2CuO
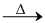 Cu2O +
Cu2O + 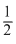 O2
O2CuSO4
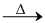 CuO + SO2 +
CuO + SO2 + 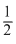 O2
O2 Create a free account to view solution
View Solution FreeMore d and f-Block Elements Questions
Cerium (Z = 58) is an important member of the lanthanoids. Which of the following statements about cerium is incorrect?...The atomic numbers of vanadium (V), Chromium (Cr), manganese (Mn) and iron (Fe) are respectively 23, 24, 25 and 26. Whic...The actinoids exhibits more number of oxidation states in general than the lanthanoids. This is because...When copper is heated with conc. HNO3 it produces:...Most common oxidation states of Ce (cerium) are...