d and f-Block ElementsHard
Question
The atomic numbers of vanadium (V), Chromium (Cr), manganese (Mn) and iron (Fe) are respectively 23, 24, 25 and 26. Which one of these may be expected to have the highest second ionization enthalpy ?
Options
A.Cr
B.Mn
C.Fe
D.V
Solution
Electronic configuration of Cr is
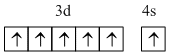
So due to half filled orbital I.P. is high of Cr.

So due to half filled orbital I.P. is high of Cr.
Create a free account to view solution
View Solution FreeMore d and f-Block Elements Questions
Glucose (A) + CO2(A) when subjected to Victor-Meyer′s test gives:-...Calamine is an ore of :...In the manufacture of iron from an iron oxide ore, limestone is added because it acts as :-...Of the following outer electronic configurations of atoms, the highest oxidation state is achieved by which one of them?...The complex entities [CoF6]3- and [Co(NH3)6]3+ differ from each other in :-...