Atomic StructureHard
Question
What is the energy of He+ electron in first orbit ?
Options
A.40.8eV
B.-27.2eV
C.-54.4eV
D.-13.6eV
Solution
E = 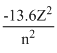 eV for He+, Z = 2, n = 1(first orbit)
eV for He+, Z = 2, n = 1(first orbit)
∴ E =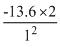 = - 54.4eV
= - 54.4eV
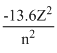 eV for He+, Z = 2, n = 1(first orbit)
eV for He+, Z = 2, n = 1(first orbit)∴ E =
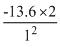 = - 54.4eV
= - 54.4eVCreate a free account to view solution
View Solution FreeMore Atomic Structure Questions
In 88Ra226 nucleus there are :...The minimum potential different between the base and emitter required to switch a silicon transistor ′ON′ is...One mole of an ideal diatomic gas undergoes a transition from A to B along a path AB as shown in the figure, The change ...When an ideal triatomic non-linear gas is heated at constant pressure, the fraction of the heat energy supplied which in...When 5V potential difference is applied across a wire of length 0.1 m, the drift speed of electrons is 2.5 × 10-4 m...