ThermodynamicsHard
Question
A monoatomic gas at pressure P1 and volume V1 is compressed adiabatically to 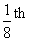 of its original volume. What is the final pressure of the gas ?
of its original volume. What is the final pressure of the gas ?
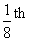 of its original volume. What is the final pressure of the gas ?
of its original volume. What is the final pressure of the gas ?Options
A.64 P1
B.P1
C.16 P1
D.32 P1
Solution
Ideal gas equation, for an abiabatic process is
PVγ = constant or P1V1γ = P2V2γ
For monoatomic gas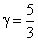
∴ P1V15/3 = P2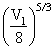 ⇒ P2 = P1 × (2)5 = 32P1
⇒ P2 = P1 × (2)5 = 32P1
PVγ = constant or P1V1γ = P2V2γ
For monoatomic gas
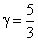
∴ P1V15/3 = P2
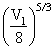 ⇒ P2 = P1 × (2)5 = 32P1
⇒ P2 = P1 × (2)5 = 32P1Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
One mole of monoatomic gas is taken round the cyclic process. Heat absorbed by gas in process AB is :-...In the isothermal expansion of a gas :...A thermodynamic system is taken through the cycle ABCDas shown in figure. Heat rejected by the gas during the cycleis...The correct formula is :...If the coefficient of Linear expansion of a solid is 0.00009. Its coefficient of volume expansion is :...