ThermodynamicsHard
Question
The change in the entropy of a an ideal gas which went through an isothermal process from an initial state (P1, V1, T) to the final state (P2, V2, T) is equal to
Options
A.zero
B.R ln T
C.R ln 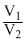
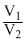
D.R ln 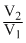
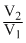
Solution
Workdone by n moles of a gas when its volume changes from V1 to V2 is,
W = nRT loge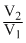
For an isothermal process, ᐃU = 0
∴ ᐃQ = DU + W = 0 + RTln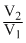
∴ Entropy =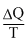 = RT ln
= RT ln 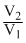
W = nRT loge
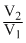
For an isothermal process, ᐃU = 0
∴ ᐃQ = DU + W = 0 + RTln
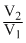
∴ Entropy =
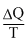 = RT ln
= RT ln 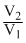
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
A bimetallic strip is formed out of two identical strips - one of copper and the other of brass. The coefficients of lin...Let and vp respectvely denote the mean speed, root mean squre speed and most monoatomic gas at absolute temperture t. Th...The value of Cp for a diatomic gas is :...In the following process, internal energy of the gas will increase : -...The molar specific heat under constant pressure of oxygen is CP = 7.03 cal/mole k. The quantity of heat required to rais...