ThermodynamicsHard
Question
Let 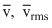 and vp respectvely denote the mean speed, root mean squre speed and most monoatomic gas at absolute temperture t. The mass of the molecule is m. Then
and vp respectvely denote the mean speed, root mean squre speed and most monoatomic gas at absolute temperture t. The mass of the molecule is m. Then
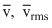 and vp respectvely denote the mean speed, root mean squre speed and most monoatomic gas at absolute temperture t. The mass of the molecule is m. Then
and vp respectvely denote the mean speed, root mean squre speed and most monoatomic gas at absolute temperture t. The mass of the molecule is m. ThenOptions
A.no molecule can have a speed greater than (√2vrms)
B.no molecule can have a speed less than 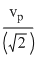
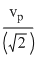
C.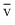 < vp < Vrms
< vp < Vrms
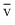 < vp < Vrms
< vp < Vrms D.The average kinetic A and B with frictionaless is 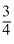 (mvp2)
(mvp2)
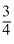 (mvp2)
(mvp2)Solution
vrms = 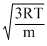
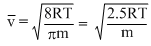
and vp =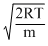
From these expressions, we can see that vp <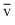 < vrms
< vrms
Again, vrms = vp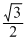
and average kinetic energy of a gas molecule Ek =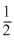 mv2rms
mv2rms
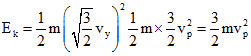
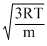
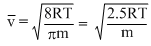
and vp =
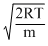
From these expressions, we can see that vp <
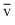 < vrms
< vrmsAgain, vrms = vp
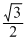
and average kinetic energy of a gas molecule Ek =
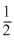 mv2rms
mv2rmsCreate a free account to view solution
View Solution FreeMore Thermodynamics Questions
In an isothermal process, the supplied heat to an ideal gas is used in :-...An insulated container of gas has two chambers separated by an insulating partition. One of the chambers has volume V1 a...For an ideal gas, each of the following processes definitely leads to an change in the pressure of the gas, except one. ...If the temperature of a body is 140oF, its value in oC will be :-...A Carnot engine, having an efficiency of η = 1/10 as heat engine, is used as a refrigerator. If the work done on th...