ThermodynamicsHard
Question
The enthalpy of fusion of water is 1.435 kcal/mol. The molar entropy change for the melting of ice at 0oC
Options
A.0.526 cal/(mol K)
B.10.52 cal / (mol K)
C.21.04 cal / (mol K)
D.5.260 cal/(mol K)
More Thermodynamics Questions
Choose the substance which has higher possible entropy (per mole) at a given temperature....A liquid is adiabatically expanded from state – I to state – II, suddenly by a single step, as shown in the figure then...If all degree of freedom of a three- dimensional N-atomic gaseous molecules is excited, then CP/CV ratio of gas should b...The work done on one mole of an ideal gas in the reversible process: PV3 = constant, from (1 atm, 300 K) to 2 2 atm is...An isolated system comprises of the liquid in equilibrium with vapours. At this stage, the molar entropy of the vapour i...
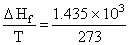 = 5.256 cal /molK
= 5.256 cal /molK