ThermodynamicsHard
Question
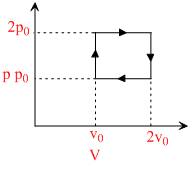
The above p-v diagram represents the thermodynamic cycle of an engine, operating with an ideal monoatomic gas. The amount of heat, extracted from the source in a single cycle is :
Options
A.p0v0
B.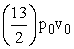
C.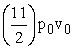
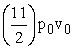
D.4p0v0
Solution
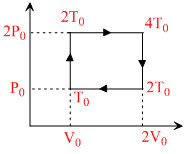
heat supplied = nCv(2T0 - T0) + nCp (4T0 - 2T0)
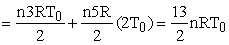
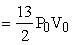
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
When an ideal diatomic gas is a heated at a constant pressure, the fraction of heat energy supplied which increases the ...$20.0{dm}^{3}$ of an ideal gas ' X ' at 600 K and 0.5 MPa undergoes isothermal reversible expansion until pressure of th...Select the correct statement...The entropy change of 2.0 moles of an ideal gas whose adiabatic exponent $\gamma$ = 1.50, if as a result of a certain pr...For a process to occur under isothermal conditions, the essential condition(s) is/ are...