KTGHard
Question
In the given (V - T) diagram, what is the relation between pressure P1 and P2 ?
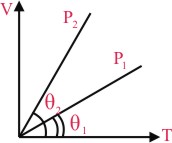

Options
A.Cannot be predicted
B.P2 = P1
C.P2 > P1
D.P2 < P1
Solution
PV = nRT ⇒ 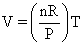 ⇒ slope
⇒ slope 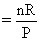
As θ2 > θ1 so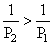 ⇒ P1 > P2
⇒ P1 > P2
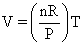 ⇒ slope
⇒ slope 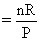
As θ2 > θ1 so
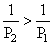 ⇒ P1 > P2
⇒ P1 > P2 Create a free account to view solution
View Solution FreeMore KTG Questions
When temperature is increased from 0oC to 273oC, in what ratio the average kinetic energy of molecules change ?...Which gas has maximum root mean square speed at same temperature and pressure conditions-...If distance between the gas molecules is doubled on constant temperature, then pressure-...Ratio of molecular specific heats of a di- atomic gas will be :-...Four curves A, B, C and D are drawn in the Fig. for a given amount of gas. The curves which represent adiabatic and isot...