Atomic StructureHard
Question
A positron is emitted from 23Na11 . The ratio of the atomic mass and atomic number of the resulting nuclide is
Options
A.22/10
B.22/11
C.23/10
D.23/12
Solution
On position emission from nucleus, proton converts into neutron therefore atomic number decreases by one but atomic mass remains constant.
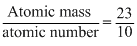
Hence (C) is correct.
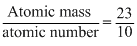
Hence (C) is correct.
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
Which of the following sets of quantum numbers represents the highest energy of an atom?...The angular momentum of an electron in a given orbit is J, Its kinetic energy will be :...The speed of electron revolving in the fourth orbit of a hydrogen-like atom or ion is 1094 km/s. The atom or ion is...If there are three possible values (–1/2, 0, +1/2) for the spin quantum number, then the maximum capacity of second orbi...Select the correct expression(s) according to Bohr model -...