Atomic StructureHard
Question
The angular momentum of an electron in a given orbit is J, Its kinetic energy will be :
Options
A.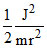
B.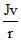
C.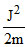
D.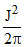
Solution
Angular momentum
J2 = m2v2r2
or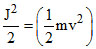 mr2 or K.E. =
mr2 or K.E. = 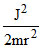
J2 = m2v2r2
or
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
Which of the following is not the possible path of cathode rays ejecting from the surface of cathode?...If the radius of first orbit of H–atom is x Å, then the radius of the second orbit of Li2+ ion will be...After np orbitals are filled, the next orbital filled will be :...What should be the increase in kinetic energy of electron in order to decrease its de Broglie wavelength from 100 nm to ...A proton and a deuteron are projected towards the stationary gold nucleus in different experiments with the same speed....