d and f-Block ElementsHard
Question
Among the following, the paramagnetic compound is
Options
A.Na2O2
B.O3
C.N2O
D.KO2
Solution
O22- = 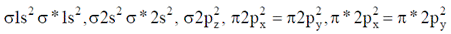
Number of unpaired electrons = 0.
N = N → O Number of unpaired electrons = 0
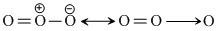 Number of unpaired electrons = 0
Number of unpaired electrons = 0
O2- =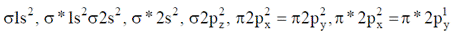
Number of unpaired electrons = 1
Thus O2- is paramagnetic.
Hence (D) is correct.
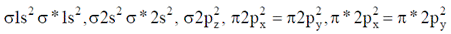
Number of unpaired electrons = 0.
N = N → O Number of unpaired electrons = 0
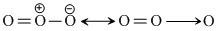 Number of unpaired electrons = 0
Number of unpaired electrons = 0 O2- =
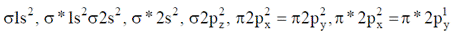
Number of unpaired electrons = 1
Thus O2- is paramagnetic.
Hence (D) is correct.
Create a free account to view solution
View Solution FreeMore d and f-Block Elements Questions
Calomel (Hg2Cl2) on reaction with ammonium hydroxide gives...A pink coloured salt turns blue on heating. The presence of which cation is most likely ?...Which one of the following nitrates will leave behind a metal on strong heating?...Which is not paramagnetic :-...Iron exhibits +2 and +3 oxidation states. Which of the following statements about iron is incorrect ?...