d and f-Block ElementsHard
Question
Iron exhibits +2 and +3 oxidation states. Which of the following statements about iron is incorrect ?
Options
A.Ferrous compounds are more easily hydrolysed than the corresponding ferric compounds.
B.Ferrous oxide is more basic in nature than the ferric oxide.
C.Ferrous compounds are relatively more ionic than the corresponding ferric compounds.
D.Ferrous compounds are less volatile than the corresponding ferric compounds.
Solution
Ferrous salt → Fe+2
Ferric salt → Fe+3
φ (Ionic potential)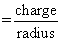
Fe+2 has low value of f compared to the Fe+3 , therefore
(1) Ferrous compounds will be less hydrolysed in comparison of Ferric.
(2) Ferrous oxide will be more basic than Ferric oxide.
(3) Ferrous compounds are relatively more ionic than Ferric compounds.
(4) Ferrous compounds have higher ionic character i.e. less volatile.
Ferric salt → Fe+3
φ (Ionic potential)
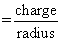
Fe+2 has low value of f compared to the Fe+3 , therefore
(1) Ferrous compounds will be less hydrolysed in comparison of Ferric.
(2) Ferrous oxide will be more basic than Ferric oxide.
(3) Ferrous compounds are relatively more ionic than Ferric compounds.
(4) Ferrous compounds have higher ionic character i.e. less volatile.
Create a free account to view solution
View Solution FreeMore d and f-Block Elements Questions
The existence of two different coloure complexes with the composition of [Co(NH3)4Cl2]+ is due to...Select the correct statements :...A red solid is insoluble in water. However it becomes soluble if some KI is added to water. Heating the red solid in a t...Dimethyl glyoxime is used for the test of ........ in alkaline medium :-...What is the correct order of spin only magnetic moment of Mn2+, Cr2+ and V2+ :-...