Ionic EquilibriumHard
Question
The conjugate base of [AI(H2O)3(OH)3] is :
Options
A.[AI(H2O)3(OH)2]+
B.[AI(H2O)3(OH)2O]-
C.[AI(H2O)3(OH)3]-
D.[AI(H2O)2(OH)4]-
Solution
Acid 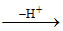 Conjugate base, Base
Conjugate base, Base 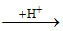 Conjugate acid
Conjugate acid
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
In a buffer solution containing equal concentration of B- and HB, the Kb for B-1 is 10-10 . The pH of buffer solution is...Let the solubility of an aqueous solution of Mg(OH)2 be x then its ksp is...What would be the pH of an ammonia solution if the pH of acetic acid solution of same strength is 3.2? The dissociation ...Which of the following will occur if a 0.1 M solution of a weak acid is diluted to 0.01 M at constant temperature?...A sample of AgCl was treated with 5.00 ml of 2.0 M Na2CO3 solution to give Ag2CO3. The remaining solution contained 0.00...