p-Block elementsHard
Question
The reaction of P4 with X leads selectively to P4O6. The X is
Options
A.Dry O2
B.A mixture of O2 and N2
C.Moist O2
D.O2 in the presence of aqueous NaOH
Solution
P4 + 3O2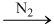 P4O6 (exclusively)
P4O6 (exclusively)
(N2 is used to retard the further oxidation.)
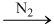 P4O6 (exclusively)
P4O6 (exclusively)(N2 is used to retard the further oxidation.)
Create a free account to view solution
View Solution FreeMore p-Block elements Questions
Non - oxide ceramics can be...Which of the following is not oxidised by MnO2 ?...Na2S2O5 + HCl → NaHSO3 + NaCl + [X] ; X is :...Antiseptics and disinfectants either kill or prevent growth of microganisms. Identify which of the following statement i...Which of the following statement(s) is/are correct ?...