Chemical EquilibriumHard
Question
For a cell reaction involving a two-electron change, the standard e.m.f. of the cell is found to be 0.295 V at 25oC. The equilibrium constant of the reaction at 25oC will be
Options
A.29.5 × 10-2
B.10
C.1 × 1010
D.1 × 10-10
Solution
The equilibrium constant is related to the standard emf of cell by the expression
log K = E0cell ×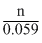 = 0.295 ×
= 0.295 × 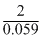
log K =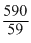 = 10 or K = 1 × 10-10
= 10 or K = 1 × 10-10
log K = E0cell ×
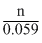 = 0.295 ×
= 0.295 × 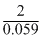
log K =
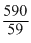 = 10 or K = 1 × 10-10
= 10 or K = 1 × 10-10Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
What will the change on increasing the pressure at equilibrium water - water vapour?...For the reaction N2O4(g)$\rightleftharpoons$ 2NO2(g), the relation between the degree of dissociation of N2O4(g) at pres...The equilibrium constant for the reaction SO3(g) ⇋ SO2(g) + O2(g) is Kc = 4.9 × 10-2. The value of Kc for the...Consider the reaction equilibrium 2SO2(g) + O2(g) ⇋ 2SO3 (g); ᐃHo = - 198 kJ. On the basis of Le Chatelier&#...The reaction A(g) + B(g) ⇋ C(g) + D(g) is studied in a one litre vessel at 250oC. The initial concentration of A w...