Redox and Equivalent ConceptHard
Question
Which of the following is a redox reaction ?
Options
A.NaCl + KNO3 → NaNO3 + KCl
B.CaC2O4 + 2HCl → CaCl + H2C2O4
C.Mg(OH)2 + 2NH4Cl → MgCl2 + 2NH4OH
D.Zn + 2AgCN → 2Ag + Zn(CN)2
Solution
The oxidation states show a change only in reaction (d)
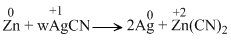
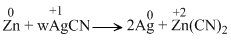
Create a free account to view solution
View Solution FreeMore Redox and Equivalent Concept Questions
The number of moles of Cr2O72− needed to oxidize 0.136 equivalent of N2H5+ through the reactionN2H5+ + Cr2O72− → N2 + Cr...A mixture of potassium chlorate, oxalic acid and sulphuric acid is heated. During the reaction which element undergoes m...A metal carbonate on heating is converted to metal oxide and is reduced to 60% of its original weight. The equivalent we...Equivalent weight of water in a neutralization reaction between dibasic acid and triacidic base is...Which of the following reaction is an example of comproportionation reaction?...