Atomic StructureHard
Question
The frequency of radiation emitted when the electron falls from n = 4 to n = 1 in a hydrogen atom will be
(Given h = 6.625 × 10-34 Js) :-
(Given h = 6.625 × 10-34 Js) :-
Options
A.3.08 × 1015 s-1
B.2.00 × 1016 s-1
C.1.54 × 1017 s-1
D.1.03 × 1014 s-1
Solution
v = 3.29 × 1015 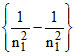 sec-1
sec-1
v = 3.29 × 1015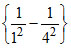
= 3.29 × 1015 ×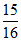 sec-1 = 3.08 × 1015 sec-1
sec-1 = 3.08 × 1015 sec-1
v = 3.29 × 1015
= 3.29 × 1015 ×
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
The sum of the number of neutrons and proton in the isotope(T) of hydrogen is :...When an electron jumps from the second orbit to fourth orbit, its distance from nucleus increases by 2.116 Å. The atom o...Because of lanthanoid contraction, which of the following pairs of elements have nearly same atomic radii ? (Nubmers in ...The following electronic transitions occur when Lithium atoms are sprayed into a hot flame;Which of these transition wou...A photon of energy hn is absorbed by a free electron of a metal having work function w < hn. Then :...