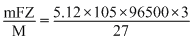ElectrochemistryHard
Question
Aluminium oxide may be electrolysed at 1000oC to furnish aluminium metal (Atomic mass = 27 amu; 1 Faraday = 96,500 Coulombs). The cathode reaction is
Al3+ + 3e- → Alo
To prepare 5.12 kg of aluminium metal by this method would require
Al3+ + 3e- → Alo
To prepare 5.12 kg of aluminium metal by this method would require
Options
A.5.49 × 107 C of electricity
B.1.83 × 107 C of electricity
C.5.49 × 104 C of electricity
D.5.49 × 101 C of electricity
More Electrochemistry Questions
Molar conductance of BaCl2, H2SO4 and HCl at infinite dilutions are x1, x2 and x3 respectively.Equivalent conductance of...Three faradays of electricity is passed through molten Al2O3, aqueous solutions of CuSO4 and molten NaCl. The amounts of...Consider the reaction of extraction of gold from its ore:Au(s) + 2CN− (aq) + ¼ O2(g) + ½ H2O(l) → Au(CN)2−(aq) + OH−(aq)...Ostwald′s dilution law is application on :...2Ce4+ + Co → 2Ce3+ + Co2+ Eocell = 1.89 V, EoCo2+/Co = - 0.277 V hence, EoCe4+Ce3+ is :...