ThermodynamicsHardBloom L3
Question
In conversion of lime-stone to lime,
CaCO3(s) → CaO(s) + CO2 (g)
the vales of ᐃHo and ᐃSo are + 179.1 kJ mol-1 and 160.2 J/K respectively at 298 K and 1 bar. Assuming that ᐃHo do not change with temperature, temperature above which conversion of limestone to lime will be spontaneous is
CaCO3(s) → CaO(s) + CO2 (g)
the vales of ᐃHo and ᐃSo are + 179.1 kJ mol-1 and 160.2 J/K respectively at 298 K and 1 bar. Assuming that ᐃHo do not change with temperature, temperature above which conversion of limestone to lime will be spontaneous is
Options
A.1008 K
B.1200
C.845 K
D.1118 K
Solution
We know, ᐃG = ᐃH - TᐃS
So, lets find the equilibrium temperature, i.e. at which
ᐃG = 0
ᐃH = TᐃS
T =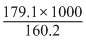 = 1118 K
= 1118 K
So, at temperature above this, the reaction will become spontaneous.
Hence, (4) is correct answer.
So, lets find the equilibrium temperature, i.e. at which
ᐃG = 0
ᐃH = TᐃS
T =
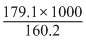 = 1118 K
= 1118 KSo, at temperature above this, the reaction will become spontaneous.
Hence, (4) is correct answer.
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
One mole of an ideal gas at 300 K is expended isothermally from an initial volume of 1 liter to 10 liters. The value of ...The enthalpy and entropy change for a chemical reaction are −2500 cal and +7.4 cal/K, respectively. The nature of reacti...Calculate the equilibrium constant for the reaction :PCl5 ⇋ PCl3 + Cl2 (g) (g) (g)at 400K if ᐃHo = 69418.86 ...The normal boiling point of water is 100°C. At 100°C...ΔS will be highest for which of the following reaction?...