Coordination CompoundHard
Question
Which one of the following has a square planar geometry?
Options
A.[CoCl4]2-
B.[FeCl4]2-
C.[NiCl4]2-
D.[PtCl4]2-
Solution
27Co2+ - 1s2 2s2 2p63s23p63d74s0
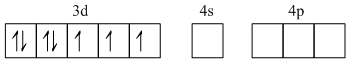
As Cl- is weak field ligand so no pairing up.
Hence it is sp3 hybridized giving tetrahedral geometry.
Fe2+ - 1s22s22p63s23p63d64s0
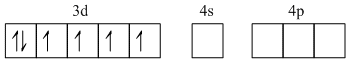
Due to Cl-, back pairing is not observed so it will be sp3 hybridized giving tetrahedral geometry.
Ni2+ - 1s22s22p63s23p63d84s0
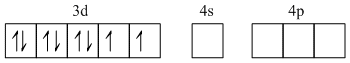
Because weak ligand, back pairing is not observed so it will be sp3 i.e. tetrahedral geometry.
All the complexes of Pt2+ are square planar including those with weak field ligand such as halide ions thus (4) is correct.

As Cl- is weak field ligand so no pairing up.
Hence it is sp3 hybridized giving tetrahedral geometry.
Fe2+ - 1s22s22p63s23p63d64s0

Due to Cl-, back pairing is not observed so it will be sp3 hybridized giving tetrahedral geometry.
Ni2+ - 1s22s22p63s23p63d84s0

Because weak ligand, back pairing is not observed so it will be sp3 i.e. tetrahedral geometry.
All the complexes of Pt2+ are square planar including those with weak field ligand such as halide ions thus (4) is correct.
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
The total number of possible coordination isomer for the given compound is[Pt(NH3)4Cl2][PtCl4]....Which of the following complexes has a geometry different from others ?...The donor sites of (EDTA)4- are ?...Which of the following complexes is not a chelate ?...Which of the following complex species is not expected to exhibit optical isomerism ?...