Coordination CompoundHard
Question
Which of the following complex species is not expected to exhibit optical isomerism ?
Options
A.[Co(en)3]3+
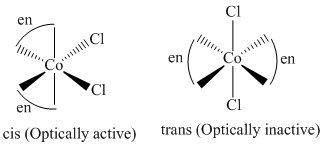
B.[Co(en)2 Cl2]+
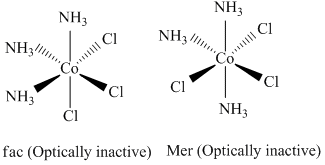
C.[Co(NH3)3 Cl3]
D.[Co(en) (NH3)2Cl2]+
Solution
(1) 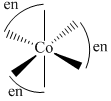 (Optically active)
(Optically active)
(2)
(3)
(4) Exist in three G.I.
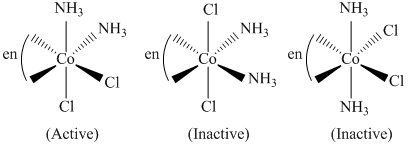
 (Optically active)
(Optically active)(2)

(3)

(4) Exist in three G.I.

Create a free account to view solution
View Solution Free