ThermodynamicsHard
Question
A gaseous mixture consists of 16 g of helium and 16 g of oxygen. The ratio 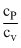 of the mixture is
of the mixture is
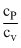 of the mixture is
of the mixture isOptions
A.1.59
B.1.62
C.1.4
D.1.54
More Thermodynamics Questions
At constant pressure wokdone in changing the volume from V1 to V2 is :-...Steam at 100o C is passes into 1.1 kg of water contained in a calorimeter of water equivalent 0.02 kg at 15oC till the t...For isometric process : -...Show below are the black body radiation curves at temperature T1 and T2 (T2 > T1). Which of the following plots is corre...Which statement is incorrect ?...
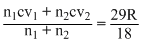
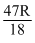 ,
, 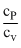 = 1.62
= 1.62