ElectrochemistryHard
Question
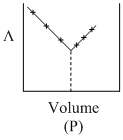
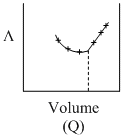
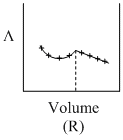
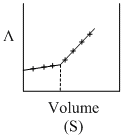
AgNO3(aq.) was added to an aqueous KCl solution gradually and the conductivity of the solution was measured. The plot of conductance (L) versus the volume of AgNO3 is




Options
A.(P)
B.(Q)
C.(R)
D.(S)
Solution
AgNO3 + KCl(aq) → AgCl(s) + KNO3 (aq)
Initially there is aq. KCl solution now as solution of AgNO3 is added, AgCl(s) is formed.
Hence conductivity of solution is almost compensated (or slightly increase) by the formation of KNO3. After end point conductivity increases more rapidly because addition of excess AgNO3 solution.
Initially there is aq. KCl solution now as solution of AgNO3 is added, AgCl(s) is formed.
Hence conductivity of solution is almost compensated (or slightly increase) by the formation of KNO3. After end point conductivity increases more rapidly because addition of excess AgNO3 solution.
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
Among the following molecules, the correct order of C - C bond lenght is...A certain current liberated 0.504 g of hydrogen in 2 hours. How many grams of copper can be liberated by the same curren...The Gibbs energy for the decomposition of Al2O3 at 500oC is as follows : Al2O3 → Al + O2, ᐃrG = + 966 kJ mol...The standard reduction potential at 25°C of the reaction 2H2O + 2e− $\rightleftharpoons$H2 + 2OH− is −0.84 V. Calculate ...Which of the following statement is incorrect :-...